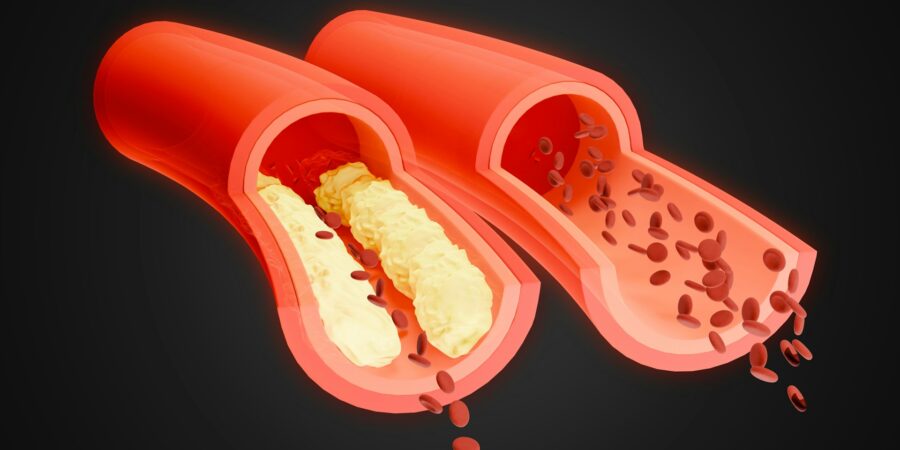
A blood clot within the artery, arterial thrombosis develops after fatty deposits collect and build up along the walls of these blood vessels. This hardens and narrows arteries in ways that obstruct or block blood flow to major organs such as the brain and heart.
In cases where arteries leading to the heart are affected, muscle pain called angina often occurs. When the blood clot is directly in the way of an artery that links to part of the heart muscle, a heart attack can result. When the clot blocks an artery that travels to the brain, stroke is likely.
The pathophysiology, as researched by physicians such as Charles V. Pollack, MD, is complex and involves blood platelets localizing at sites where atherosclerotic plaque collects. Interactions between collagen and glycoprotein Ib and VI (GPIb and GPVI) on the platelet surface build molecular bonds.
When tethered as part of a thrombus (coagulating mass), platelets release the signaling molecule Adenosine diphosphate (ADP) and thromboxane A2 (TXA2). These activate ambient platelets, which aggregate through a process of activated GPIIb/IIIa. They bind together with fibrinogen, which bridges the various platelets. In addition, tissue factor coagulation at the injury site triggers thrombin production, which induces more fibrin formation, in a progressive process that narrows the artery.
Another important element is coagulation factor XI (FXI). The thrombus (solid blood clot) at the plaque disruption site quickly covers the injury, which prevents activation of tissue factor–mediated coagulation. FXI now takes the lead in encouraging fibrin formation and thrombus expansion.
A next generation of anticoagulants are actually targeting FXI. These are safer than the present class of available anticoagulants, as FXI is not essential to hemostasis, or the multi-stage process of stopping vascular bleeding.
One minimally invasive way of treating blood clot is through percutaneous coronary intervention (PCI). This involves using catheterization and stent placement to open blocked arteries and restore proper blood flow.
Following PCI, standard treatment involves aspirin and a P2Y12 inhibitor. This dual antiplatelet therapy (DAPT) approach addresses risks of recurrent ischemic events and stent thrombosis. While traditional DAPT lasted 12 months or longer, the duration of DAPT today is often much shorter (depending on bleeding and ischemia risks). Ischemia involves reduced oxygen and blood flow to tissues, which causes clots and arterial blockages.
With next-generation drug-eluting stents in place, late stent thrombosis risks are mitigated. The makes extended DAPT treatment less necessary. Patients of physicians such as Charles V. Pollack, MD, with a combination of low—ischemic-event and high bleeding risk, may benefit from a DAPT regimen lasting only one to three months.
Physicians are moving away from aspirin as a foundation of secondary prevention. In one trial of atrial fibrillation patients, stopping aspirin a month into DAPT helped decrease bleeding risks, with no efficacy issues. Subsequent studies found that, after one to three months of DAPT, taking a P2Y12 inhibitor, rather than aspirin, decreases bleeding and ischemic risks. Some physicians also recommend using weaker P2Y12 inhibitors such as clopidogrel, instead of stronger inhibitors such as ticagrelor and prasugrel. The basic philosophy of physicians such as Charles V. Pollack, MD, is to recommend the minimum, least-risky therapy to get the job done.